Journal Description
Cells
Cells
is an international, peer-reviewed, open access journal on cell biology, molecular biology, and biophysics, published semimonthly online by MDPI. The Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH) and Society for Regenerative Medicine (Russian Federation) (RPO) are affiliated with Cells and their members receive discounts on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, MEDLINE, PMC, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Cell Biology) / CiteScore - Q1 (General Biochemistry, Genetics and Molecular Biology)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 16.6 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Sections: published in 21 topical sections.
- Companion journal: Organoids.
Impact Factor:
6.0 (2022);
5-Year Impact Factor:
6.7 (2022)
Latest Articles
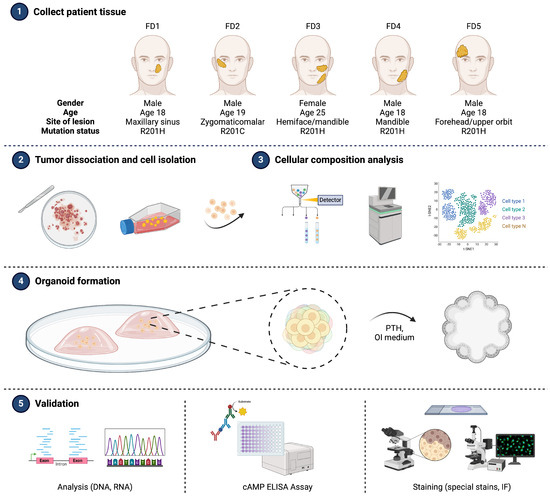
Patient-Derived Organoids Recapitulate Pathological Intrinsic and Phenotypic Features of Fibrous Dysplasia
Cells 2024, 13(9), 729; https://doi.org/10.3390/cells13090729 (registering DOI) - 23 Apr 2024
Abstract
Fibrous dysplasia (FD) is a rare bone disorder characterized by the replacement of normal bone with benign fibro-osseous tissue. Developments in our understanding of the pathophysiology and treatment options are impeded by the lack of suitable research models. In this study, we developed
[...] Read more.
Fibrous dysplasia (FD) is a rare bone disorder characterized by the replacement of normal bone with benign fibro-osseous tissue. Developments in our understanding of the pathophysiology and treatment options are impeded by the lack of suitable research models. In this study, we developed an in vitro organotypic model capable of recapitulating key intrinsic and phenotypic properties of FD. Initially, transcriptomic profiling of individual cells isolated from patient lesional tissues unveiled intralesional molecular and cellular heterogeneity. Leveraging these insights, we established patient-derived organoids (PDOs) using primary cells obtained from patient FD lesions. Evaluation of PDOs demonstrated preservation of fibrosis-associated constituent cell types and transcriptional signatures observed in FD lesions. Additionally, PDOs retained distinct constellations of genomic and metabolic alterations characteristic of FD. Histological evaluation further corroborated the fidelity of PDOs in recapitulating important phenotypic features of FD that underscore their pathophysiological relevance. Our findings represent meaningful progress in the field, as they open up the possibility for in vitro modeling of rare bone lesions in a three-dimensional context and may signify the first step towards creating a personalized platform for research and therapeutic studies.
Full article
(This article belongs to the Special Issue Model Systems for Human Disease and Medicine: From Advanced Cell and Tissue Culture to In Silico Methods)
►
Show Figures
Open AccessArticle
Activin A, a Novel Chemokine, Induces Mouse NK Cell Migration via AKT and Calcium Signaling
by
Yunfeng Wang, Zhonghui Liu, Yan Qi, Jiandong Wu, Boyang Liu and Xueling Cui
Cells 2024, 13(9), 728; https://doi.org/10.3390/cells13090728 (registering DOI) - 23 Apr 2024
Abstract
Natural killer (NK) cells can migrate quickly to the tumor site to exert cytotoxic effects on tumors, and some chemokines, including CXCL8, CXCL10 or and CXCL12, can regulate the migration of NK cells. Activin A, a member of the transforming growth factor β
[...] Read more.
Natural killer (NK) cells can migrate quickly to the tumor site to exert cytotoxic effects on tumors, and some chemokines, including CXCL8, CXCL10 or and CXCL12, can regulate the migration of NK cells. Activin A, a member of the transforming growth factor β (TGF-β) superfamily, is highly expressed in tumor tissues and involved in tumor development and immune cell activation. In this study, we focus on the effects of activin A on NK cell migration. In vitro, activin A induced NK cell migration and invasion, promoted cell polarization and inhibited cell adhesion. Moreover, activin A increased Ca2+, p-SMAD3 and p-AKT levels in NK cells. An AKT inhibitor and Ca2+ chelator partially blocked activin A-induced NK cell migration. In vivo, exogenous activin A increased tumor-infiltrating NK cells in NS-1 cell solid tumors and inhibited tumor growth, and blocking endogenous activin A with anti-activin A antibody reduced tumor-infiltrating NK cells in 4T-1 cell solid tumors. These results suggest that activin A induces NK cell migration through AKT signaling and calcium signaling and may enhance the antitumor effect of NK cells by increasing tumor-infiltrating NK cells.
Full article
(This article belongs to the Section Cellular Immunology)
►▼
Show Figures
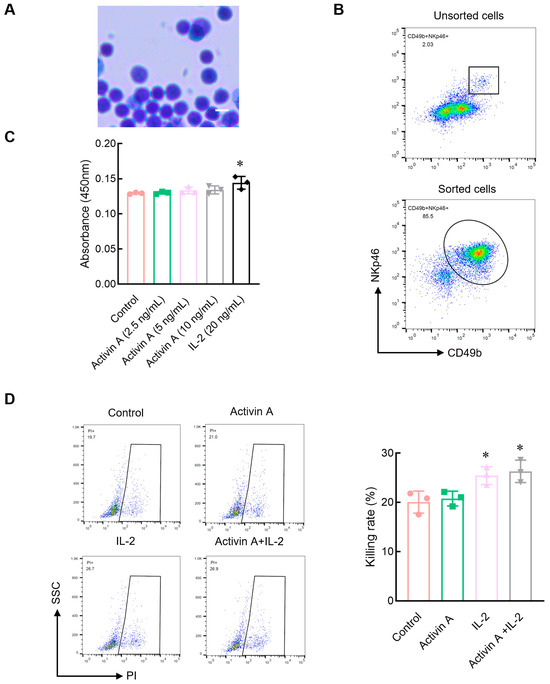
Figure 1
Open AccessReview
Copper Homeostasis in the Model Organism C. elegans
by
Verena Alexia Ohse, Lars-Oliver Klotz and Josephine Priebs
Cells 2024, 13(9), 727; https://doi.org/10.3390/cells13090727 (registering DOI) - 23 Apr 2024
Abstract
Cellular and organismic copper (Cu) homeostasis is regulated by Cu transporters and Cu chaperones to ensure the controlled uptake, distribution and export of Cu ions. Many of these processes have been extensively investigated in mammalian cell culture, as well as in humans and
[...] Read more.
Cellular and organismic copper (Cu) homeostasis is regulated by Cu transporters and Cu chaperones to ensure the controlled uptake, distribution and export of Cu ions. Many of these processes have been extensively investigated in mammalian cell culture, as well as in humans and in mammalian model organisms. Most of the human genes encoding proteins involved in Cu homeostasis have orthologs in the model organism, Caenorhabditis elegans (C. elegans). Starting with a compilation of human Cu proteins and their orthologs, this review presents an overview of Cu homeostasis in C. elegans, comparing it to the human system, thereby establishing the basis for an assessment of the suitability of C. elegans as a model to answer mechanistic questions relating to human Cu homeostasis.
Full article
(This article belongs to the Special Issue Caenorhabditis elegans: A Model Organism, Endless Possibilities)
►▼
Show Figures
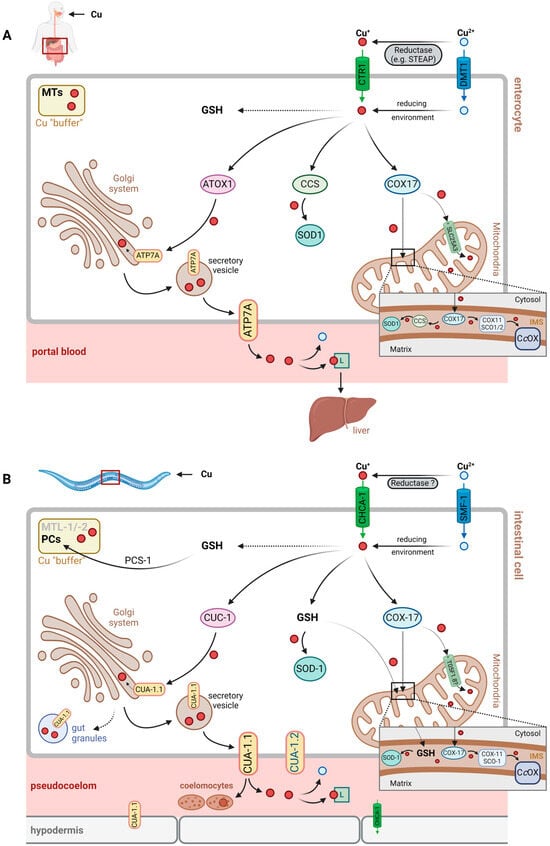
Figure 1
Open AccessReview
A Roadmap of CAR-T-Cell Therapy in Glioblastoma: Challenges and Future Perspectives
by
Megan Montoya, Marco Gallus, Su Phyu, Jeffrey Haegelin, John de Groot and Hideho Okada
Cells 2024, 13(9), 726; https://doi.org/10.3390/cells13090726 - 23 Apr 2024
Abstract
Glioblastoma (GBM) is the most common primary malignant brain tumor, with a median overall survival of less than 2 years and a nearly 100% mortality rate under standard therapy that consists of surgery followed by combined radiochemotherapy. Therefore, new therapeutic strategies are urgently
[...] Read more.
Glioblastoma (GBM) is the most common primary malignant brain tumor, with a median overall survival of less than 2 years and a nearly 100% mortality rate under standard therapy that consists of surgery followed by combined radiochemotherapy. Therefore, new therapeutic strategies are urgently needed. The success of chimeric antigen receptor (CAR) T cells in hematological cancers has prompted preclinical and clinical investigations into CAR-T-cell treatment for GBM. However, recent trials have not demonstrated any major success. Here, we delineate existing challenges impeding the effectiveness of CAR-T-cell therapy for GBM, encompassing the cold (immunosuppressive) microenvironment, tumor heterogeneity, T-cell exhaustion, local and systemic immunosuppression, and the immune privilege inherent to the central nervous system (CNS) parenchyma. Additionally, we deliberate on the progress made in developing next-generation CAR-T cells and novel innovative approaches, such as low-intensity pulsed focused ultrasound, aimed at surmounting current roadblocks in GBM CAR-T-cell therapy.
Full article
(This article belongs to the Special Issue Cell and Immune Therapeutics for Gliomas)
►▼
Show Figures

Figure 1
Open AccessReview
State of the Art in CAR-T Cell Therapy for Solid Tumors: Is There a Sweeter Future?
by
Beatriz Amorós-Pérez, Benigno Rivas-Pardo, Manuel Gómez del Moral, José Luis Subiza and Eduardo Martínez-Naves
Cells 2024, 13(9), 725; https://doi.org/10.3390/cells13090725 - 23 Apr 2024
Abstract
Chimeric antigen receptor (CAR)-T cell therapy has proven to be a powerful treatment for hematological malignancies. The situation is very different in the case of solid tumors, for which no CAR-T-based therapy has yet been approved. There are many factors contributing to the
[...] Read more.
Chimeric antigen receptor (CAR)-T cell therapy has proven to be a powerful treatment for hematological malignancies. The situation is very different in the case of solid tumors, for which no CAR-T-based therapy has yet been approved. There are many factors contributing to the absence of response in solid tumors to CAR-T cells, such as the immunosuppressive tumor microenvironment (TME), T cell exhaustion, or the lack of suitable antigen targets, which should have a stable and specific expression on tumor cells. Strategies being developed to improve CAR-T-based therapy for solid tumors include the use of new-generation CARs such as TRUCKs or bi-specific CARs, the combination of CAR therapy with chemo- or radiotherapy, the use of checkpoint inhibitors, and the use of oncolytic viruses. Furthermore, despite the scarcity of targets, a growing number of phase I/II clinical trials are exploring new solid-tumor-associated antigens. Most of these antigens are of a protein nature; however, there is a clear potential in identifying carbohydrate-type antigens associated with tumors, or carbohydrate and proteoglycan antigens that emerge because of aberrant glycosylations occurring in the context of tumor transformation.
Full article
(This article belongs to the Special Issue Advancements in Cancer Immunotherapy beyond Checkpoint Blockade)
►▼
Show Figures
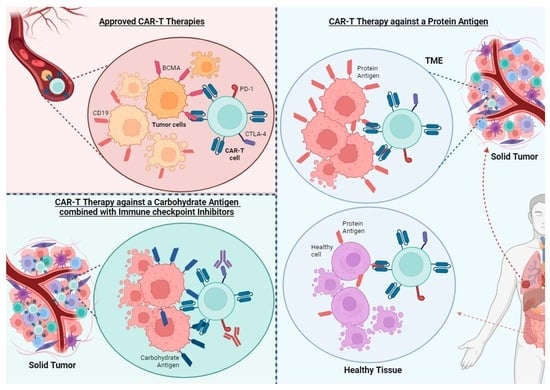
Graphical abstract
Open AccessReview
Bone Marrow Adipose Tissue
by
Elena Marinelli Busilacchi, Erika Morsia and Antonella Poloni
Cells 2024, 13(9), 724; https://doi.org/10.3390/cells13090724 - 23 Apr 2024
Abstract
Bone marrow (BM) acts as a dynamic organ within the bone cavity, responsible for hematopoiesis, skeletal remodeling, and immune system control. Bone marrow adipose tissue (BMAT) was long simply considered a filler of space, but now it is known that it instead constitutes
[...] Read more.
Bone marrow (BM) acts as a dynamic organ within the bone cavity, responsible for hematopoiesis, skeletal remodeling, and immune system control. Bone marrow adipose tissue (BMAT) was long simply considered a filler of space, but now it is known that it instead constitutes an essential element of the BM microenvironment that participates in homeostasis, influences bone health and bone remodeling, alters hematopoietic stem cell functions, contributes to the commitment of mesenchymal stem cells, provides effects to immune homeostasis and defense against infections, and participates in energy metabolism and inflammation. BMAT has emerged as a significant contributor to the development and progression of various diseases, shedding light on its complex relationship with health. Notably, BMAT has been implicated in metabolic disorders, hematological malignancies, and skeletal conditions. BMAT has been shown to support the proliferation of tumor cells in acute myeloid leukemia and niche adipocytes have been found to protect cancer cells against chemotherapy, contributing to treatment resistance. Moreover, BMAT’s impact on bone density and remodeling can lead to conditions like osteoporosis, where high levels of BMAT are inversely correlated with bone mineral density, increasing the risk of fractures. BMAT has also been associated with diabetes, obesity, and anorexia nervosa, with varying effects on individuals depending on their weight and health status. Understanding the interaction between adipocytes and different diseases may lead to new therapeutic strategies.
Full article
(This article belongs to the Special Issue Molecular Mechanisms of Adipose Organ Remodelling)
►▼
Show Figures
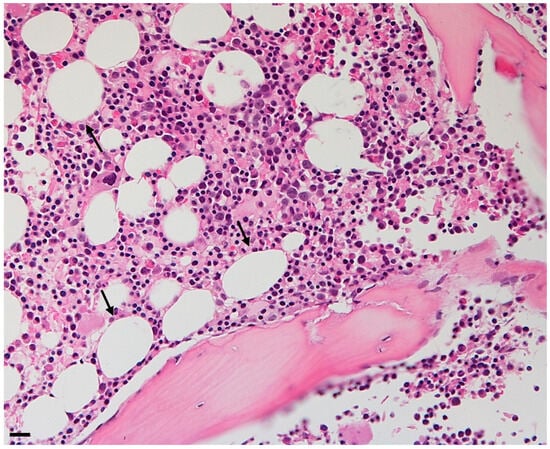
Figure 1
Open AccessArticle
High Level of CD8+PD-1+ Cells in Patients with Chronic Myeloid Leukemia Who Experienced Loss of MMR after Imatinib Discontinuation
by
Paulina Kwaśnik, Joanna Zaleska, Dorota Link-Lenczowska, Magdalena Zawada, Hubert Wysogląd, Bogdan Ochrem, Grażyna Bober, Ewa Wasilewska, Iwona Hus, Monika Szarejko, Witold Prejzner, Olga Grzybowska-Izydorczyk, Agnieszka Klonowska-Szymczyk, Ewa Mędraś, Michał Kiełbus, Tomasz Sacha and Krzysztof Giannopoulos
Cells 2024, 13(8), 723; https://doi.org/10.3390/cells13080723 - 22 Apr 2024
Abstract
Treatment-free remission (TFR) is achieved in approximately half of chronic myeloid leukemia (CML) patients treated with tyrosine kinase inhibitors. The mechanisms responsible for TFR maintenance remain elusive. This study aimed to identify immune markers responsible for the control of residual CML cells early
[...] Read more.
Treatment-free remission (TFR) is achieved in approximately half of chronic myeloid leukemia (CML) patients treated with tyrosine kinase inhibitors. The mechanisms responsible for TFR maintenance remain elusive. This study aimed to identify immune markers responsible for the control of residual CML cells early in the TFR (at 3 months), which may be the key to achieving long-term TFR and relapse-free survival (RFS) after discontinuation of imatinib. Our study included 63 CML patients after imatinib discontinuation, in whom comprehensive analysis of changes in the immune system was performed by flow cytometry, and changes in the BCR::ABL1 transcript levels were assessed by RQ-PCR and ddPCR. We demonstrated a significant increase in the percentage of CD8+PD-1+ cells in patients losing TFR. The level of CD8+PD-1+ cells is inversely related to the duration of treatment and incidence of deep molecular response (DMR) before discontinuation. Analysis of the ROC curve showed that the percentage of CD8+PD-1+ cells may be a significant factor in early molecular recurrence. Interestingly, at 3 months of TFR, patients with the e13a2 transcript had a significantly higher proportion of the PD-1-expressing immune cells compared to patients with the e14a2. Our results suggest the important involvement of CD8+PD-1+ cells in the success of TFR and may help in identifying a group of patients who could successfully discontinue imatinib.
Full article
(This article belongs to the Collection Trends and Advances in Tumor Immunology)
►▼
Show Figures
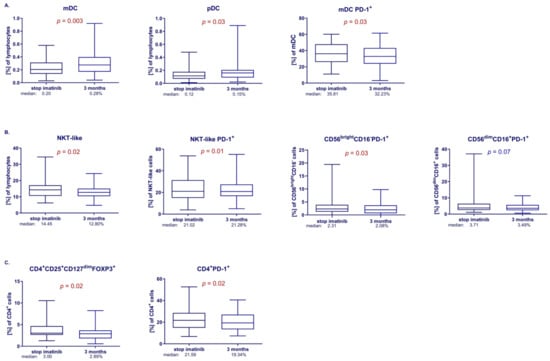
Figure 1
Open AccessCorrection
Correction: Dastghaib et al. Simvastatin Induces Unfolded Protein Response and Enhances Temozolomide-Induced Cell Death in Glioblastoma Cells. Cells 2020, 9, 2339
by
Sanaz Dastghaib, Shahla Shojaei, Zohreh Mostafavi-Pour, Pawan Sharma, John B. Patterson, Afshin Samali, Pooneh Mokarram and Saeid Ghavami
Cells 2024, 13(8), 722; https://doi.org/10.3390/cells13080722 - 22 Apr 2024
Abstract
►▼
Show Figures
In the original publication [...]
Full article

Figure 4
Open AccessReview
Drosophila Contributions towards Understanding Neurofibromatosis 1
by
Kalliopi Atsoniou, Eleni Giannopoulou, Eirini-Maria Georganta and Efthimios M. C. Skoulakis
Cells 2024, 13(8), 721; https://doi.org/10.3390/cells13080721 - 21 Apr 2024
Abstract
Neurofibromatosis 1 (NF1) is a multisymptomatic disorder with highly variable presentations, which include short stature, susceptibility to formation of the characteristic benign tumors known as neurofibromas, intense freckling and skin discoloration, and cognitive deficits, which characterize most children with the condition. Attention deficits
[...] Read more.
Neurofibromatosis 1 (NF1) is a multisymptomatic disorder with highly variable presentations, which include short stature, susceptibility to formation of the characteristic benign tumors known as neurofibromas, intense freckling and skin discoloration, and cognitive deficits, which characterize most children with the condition. Attention deficits and Autism Spectrum manifestations augment the compromised learning presented by most patients, leading to behavioral problems and school failure, while fragmented sleep contributes to chronic fatigue and poor quality of life. Neurofibromin (Nf1) is present ubiquitously during human development and postnatally in most neuronal, oligodendrocyte, and Schwann cells. Evidence largely from animal models including Drosophila suggests that the symptomatic variability may reflect distinct cell-type-specific functions of the protein, which emerge upon its loss, or mutations affecting the different functional domains of the protein. This review summarizes the contributions of Drosophila in modeling multiple NF1 manifestations, addressing hypotheses regarding the cell-type-specific functions of the protein and exploring the molecular pathways affected upon loss of the highly conserved fly homolog dNf1. Collectively, work in this model not only has efficiently and expediently modelled multiple aspects of the condition and increased understanding of its behavioral manifestations, but also has led to pharmaceutical strategies towards their amelioration.
Full article
(This article belongs to the Special Issue Drosophila Models of Development and Disease)
►▼
Show Figures
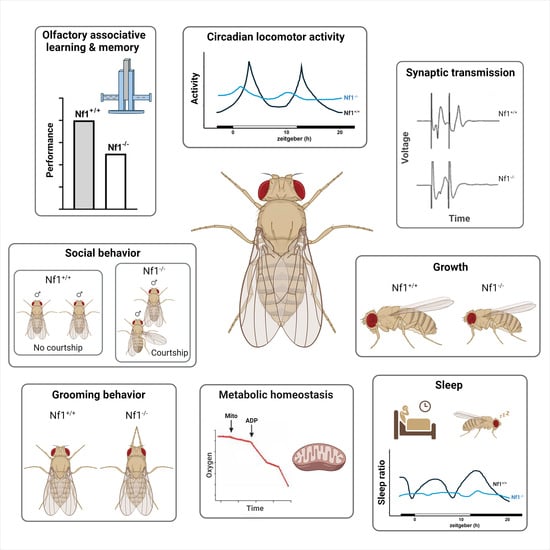
Graphical abstract
Open AccessArticle
Expression of PDLIM5 Spliceosomes and Regulatory Functions on Myogenesis in Pigs
by
Yu Fu, Shixin Li, Jingru Nie, Dawei Yan, Bo Zhang, Xin Hao and Hao Zhang
Cells 2024, 13(8), 720; https://doi.org/10.3390/cells13080720 - 21 Apr 2024
Abstract
►▼
Show Figures
Meat yield, determined by muscle growth and development, is an important economic trait for the swine industry and a focus of research in animal genetics and breeding. PDZ and LIM domain 5 (PDLIM5) are cytoskeleton-related proteins that play key roles in various tissues
[...] Read more.
Meat yield, determined by muscle growth and development, is an important economic trait for the swine industry and a focus of research in animal genetics and breeding. PDZ and LIM domain 5 (PDLIM5) are cytoskeleton-related proteins that play key roles in various tissues and cells. These proteins have multiple isoforms, primarily categorized as short (PDLIM5-short) and long (PDLIM5-long) types, distinguished by the absence and presence of an LIM domain, respectively. However, the expression patterns of swine PDLIM5 isoforms and their regulation during porcine skeletal muscle development remain largely unexplored. We observed that PDLIM5-long was expressed at very low levels in pig muscles and that PDLIM5-short and total PDLIM5 were highly expressed in the muscles of slow-growing pigs, suggesting that PDLIM5-short, the dominant transcript in pigs, is associated with a slow rate of muscle growth. PDLIM5-short suppressed myoblast proliferation and myogenic differentiation in vitro. We also identified two single nucleotide polymorphisms (−258 A > T and −191 T > G) in the 5′ flanking region of PDLIM5, which influenced the activity of the promoter and were associated with muscle growth rate in pigs. In summary, we demonstrated that PDLIM5-short negatively regulates myoblast proliferation and differentiation, providing a theoretical basis for improving pig breeding programs.
Full article
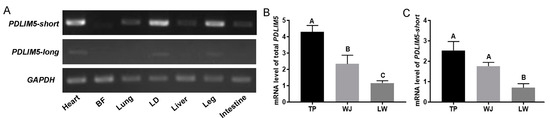
Figure 1
Open AccessReview
Epigenetic Changes in Alzheimer’s Disease: DNA Methylation and Histone Modification
by
Laura Maria De Plano, Alessandra Saitta, Salvatore Oddo and Antonella Caccamo
Cells 2024, 13(8), 719; https://doi.org/10.3390/cells13080719 - 21 Apr 2024
Abstract
►▼
Show Figures
Alzheimer’s disease (AD) is a devastating neurodegenerative disorder characterized by progressive cognitive decline and memory loss, imposing a significant burden on affected individuals and their families. Despite the recent promising progress in therapeutic approaches, more needs to be done to understand the intricate
[...] Read more.
Alzheimer’s disease (AD) is a devastating neurodegenerative disorder characterized by progressive cognitive decline and memory loss, imposing a significant burden on affected individuals and their families. Despite the recent promising progress in therapeutic approaches, more needs to be done to understand the intricate molecular mechanisms underlying the development and progression of AD. Growing evidence points to epigenetic changes as playing a pivotal role in the pathogenesis of the disease. The dynamic interplay between genetic and environmental factors influences the epigenetic landscape in AD, altering gene expression patterns associated with key pathological events associated with disease pathogenesis. To this end, epigenetic alterations not only impact the expression of genes implicated in AD pathogenesis but also contribute to the dysregulation of crucial cellular processes, including synaptic plasticity, neuroinflammation, and oxidative stress. Understanding the complex epigenetic mechanisms in AD provides new avenues for therapeutic interventions. This review comprehensively examines the role of DNA methylation and histone modifications in the context of AD. It aims to contribute to a deeper understanding of AD pathogenesis and facilitate the development of targeted therapeutic strategies.
Full article
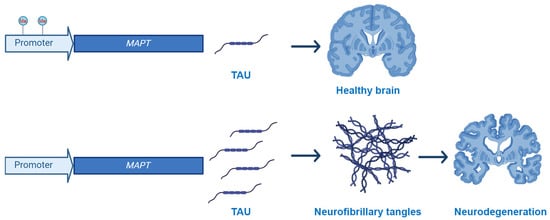
Figure 1
Open AccessArticle
AAV-Mediated Restoration of Dystrophin-Dp71 in the Brain of Dp71-Null Mice: Molecular, Cellular and Behavioral Outcomes
by
Ophélie Vacca, Faouzi Zarrouki, Charlotte Izabelle, Mehdi Belmaati Cherkaoui, Alvaro Rendon, Deniz Dalkara and Cyrille Vaillend
Cells 2024, 13(8), 718; https://doi.org/10.3390/cells13080718 - 20 Apr 2024
Abstract
A deficiency in the shortest dystrophin-gene product, Dp71, is a pivotal aggravating factor for intellectual disabilities in Duchenne muscular dystrophy (DMD). Recent advances in preclinical research have achieved some success in compensating both muscle and brain dysfunctions associated with DMD, notably using exon
[...] Read more.
A deficiency in the shortest dystrophin-gene product, Dp71, is a pivotal aggravating factor for intellectual disabilities in Duchenne muscular dystrophy (DMD). Recent advances in preclinical research have achieved some success in compensating both muscle and brain dysfunctions associated with DMD, notably using exon skipping strategies. However, this has not been studied for distal mutations in the DMD gene leading to Dp71 loss. In this study, we aimed to restore brain Dp71 expression in the Dp71-null transgenic mouse using an adeno-associated virus (AAV) administrated either by intracardiac injections at P4 (ICP4) or by bilateral intracerebroventricular (ICV) injections in adults. ICP4 delivery of the AAV9-Dp71 vector enabled the expression of 2 to 14% of brain Dp71, while ICV delivery enabled the overexpression of Dp71 in the hippocampus and cortex of adult mice, with anecdotal expression in the cerebellum. The restoration of Dp71 was mostly located in the glial endfeet that surround capillaries, and it was associated with partial localization of Dp71-associated proteins, α1-syntrophin and AQP4 water channels, suggesting proper restoration of a scaffold of proteins involved in blood–brain barrier function and water homeostasis. However, this did not result in significant improvements in behavioral disturbances displayed by Dp71-null mice. The potential and limitations of this AAV-mediated strategy are discussed. This proof-of-concept study identifies key molecular markers to estimate the efficiencies of Dp71 rescue strategies and opens new avenues for enhancing gene therapy targeting cognitive disorders associated with a subgroup of severely affected DMD patients.
Full article
(This article belongs to the Topic Animal Models of Human Disease 2.0)
►▼
Show Figures
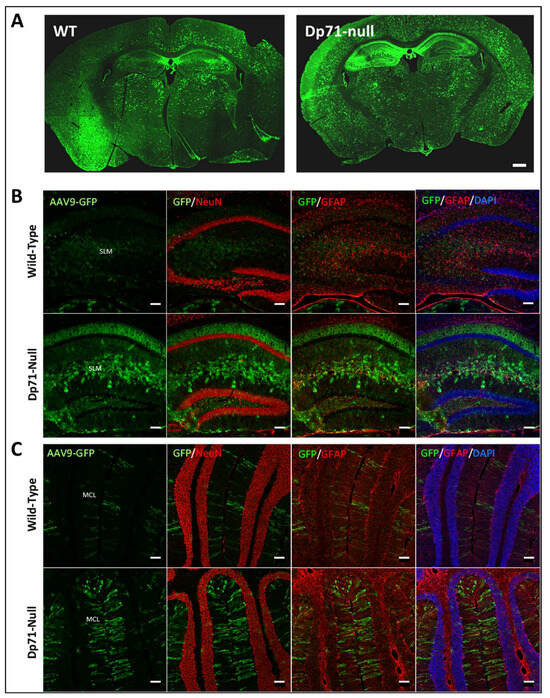
Figure 1
Open AccessReview
Fusobacterium nucleatum: An Overview of Evidence, Demi-Decadal Trends, and Its Role in Adverse Pregnancy Outcomes and Various Gynecological Diseases, including Cancers
by
Arunita Ghosh, Ken Jaaback, Angela Boulton, Michelle Wong-Brown, Steve Raymond, Partha Dutta, Nikola A. Bowden and Arnab Ghosh
Cells 2024, 13(8), 717; https://doi.org/10.3390/cells13080717 - 20 Apr 2024
Abstract
Gynecological and obstetric infectious diseases are crucial to women’s health. There is growing evidence that links the presence of Fusobacterium nucleatum (F. nucleatum), an anaerobic oral commensal and potential periodontal pathogen, to the development and progression of various human diseases, including
[...] Read more.
Gynecological and obstetric infectious diseases are crucial to women’s health. There is growing evidence that links the presence of Fusobacterium nucleatum (F. nucleatum), an anaerobic oral commensal and potential periodontal pathogen, to the development and progression of various human diseases, including cancers. While the role of this opportunistic oral pathogen has been extensively studied in colorectal cancer in recent years, research on its epidemiological evidence and mechanistic link to gynecological diseases (GDs) is still ongoing. Thus, the present review, which is the first of its kind, aims to undertake a comprehensive and critical reappraisal of F. nucleatum, including the genetics and mechanistic role in promoting adverse pregnancy outcomes (APOs) and various GDs, including cancers. Additionally, this review discusses new conceptual advances that link the immunomodulatory role of F. nucleatum to the development and progression of breast, ovarian, endometrial, and cervical carcinomas through the activation of various direct and indirect signaling pathways. However, further studies are needed to explore and elucidate the highly dynamic process of host–F. nucleatum interactions and discover new pathways, which will pave the way for the development of better preventive and therapeutic strategies against this pathobiont.
Full article
(This article belongs to the Section Cellular Pathology)
►▼
Show Figures
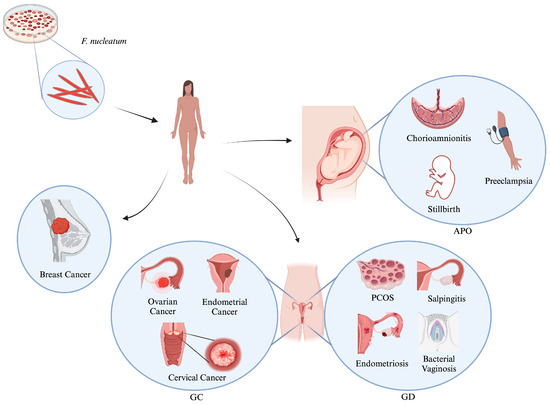
Figure 1
Open AccessArticle
The Expression and Secretion Profile of TRAP5 Isoforms in Gaucher Disease
by
Margarita M. Ivanova, Julia Dao, Neala Loynab, Sohailla Noor, Neil Kasaci, Andrew Friedman and Ozlem Goker-Alpan
Cells 2024, 13(8), 716; https://doi.org/10.3390/cells13080716 - 20 Apr 2024
Abstract
Background: Gaucher disease (GD) is caused by glucocerebrosidase (GCase) enzyme deficiency, leading to glycosylceramide (Gb-1) and glucosylsphingosine (Lyso-Gb-1) accumulation. The pathological hallmark for GD is an accumulation of large macrophages called Gaucher cells (GCs) in the liver, spleen, and bone marrow, which are
[...] Read more.
Background: Gaucher disease (GD) is caused by glucocerebrosidase (GCase) enzyme deficiency, leading to glycosylceramide (Gb-1) and glucosylsphingosine (Lyso-Gb-1) accumulation. The pathological hallmark for GD is an accumulation of large macrophages called Gaucher cells (GCs) in the liver, spleen, and bone marrow, which are associated with chronic organ enlargement, bone manifestations, and inflammation. Tartrate-resistant acid phosphatase type 5 (TRAP5 protein, ACP5 gene) has long been a nonspecific biomarker of macrophage/GCs activation; however, the discovery of two isoforms of TRAP5 has expanded its significance. The discovery of TRAP5′s two isoforms revealed that it is more than just a biomarker of macrophage activity. While TRAP5a is highly expressed in macrophages, TRAP5b is secreted by osteoclasts. Recently, we have shown that the elevation of TRAP5b in plasma is associated with osteoporosis in GD. However, the role of TRAP isoforms in GD and how the accumulation of Gb-1 and Lyso-Gb-1 affects TRAP expression is unknown. Methods: 39 patients with GD were categorized into cohorts based on bone mineral density (BMD). TRAP5a and TRAP5b plasma levels were quantified by ELISA. ACP5 mRNA was estimated using RT-PCR. Results: An increase in TRAP5b was associated with reduced BMD and correlated with Lyso-Gb-1 and immune activator chemokine ligand 18 (CCL18). In contrast, the elevation of TRAP5a correlated with chitotriosidase activity in GD. Lyso-Gb-1 and plasma seemed to influence the expression of ACP5 in macrophages. Conclusions: As an early indicator of BMD alteration, measurement of circulating TRAP5b is a valuable tool for assessing osteopenia–osteoporosis in GD, while TRAP5a serves as a biomarker of macrophage activation in GD. Understanding the distinct expression pattern of TRAP5 isoforms offers valuable insight into both bone disease and the broader implications for immune system activation in GD.
Full article
(This article belongs to the Topic Osteoimmunology and Bone Biology)
►▼
Show Figures
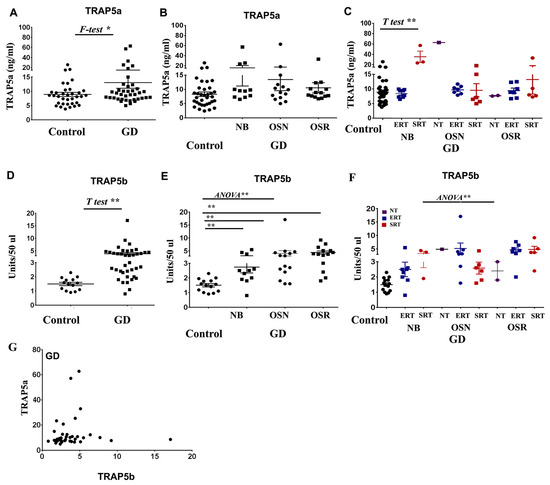
Figure 1
Open AccessArticle
5-Hydroxymethylcytosine in Cell-Free DNA Predicts Immunotherapy Response in Lung Cancer
by
Jianming Shao, Yitian Xu, Randall J. Olsen, Saro Kasparian, Kai Sun, Sunil Mathur, Jun Zhang, Chuan He, Shu-Hsia Chen, Eric H. Bernicker and Zejuan Li
Cells 2024, 13(8), 715; https://doi.org/10.3390/cells13080715 - 19 Apr 2024
Abstract
►▼
Show Figures
Immune checkpoint inhibitors (ICIs) drastically improve therapeutic outcomes for lung cancer, but accurately predicting individual patient responses to ICIs remains a challenge. We performed the genome-wide profiling of 5-hydroxymethylcytosine (5hmC) in 85 plasma cell-free DNA (cfDNA) samples from lung cancer patients and developed
[...] Read more.
Immune checkpoint inhibitors (ICIs) drastically improve therapeutic outcomes for lung cancer, but accurately predicting individual patient responses to ICIs remains a challenge. We performed the genome-wide profiling of 5-hydroxymethylcytosine (5hmC) in 85 plasma cell-free DNA (cfDNA) samples from lung cancer patients and developed a 5hmC signature that was significantly associated with progression-free survival (PFS). We built a 5hmC predictive model to quantify the 5hmC level and validated the model in the validation, test, and control sets. Low weighted predictive scores (wp-scores) were significantly associated with a longer PFS compared to high wp-scores in the validation [median 7.6 versus 1.8 months; p = 0.0012; hazard ratio (HR) 0.12; 95% confidence interval (CI), 0.03–0.54] and test (median 14.9 versus 3.3 months; p = 0.00074; HR 0.10; 95% CI, 0.02–0.50) sets. Objective response rates in patients with a low or high wp-score were 75.0% (95% CI, 42.8–94.5%) versus 0.0% (95% CI, 0.0–60.2%) in the validation set (p = 0.019) and 80.0% (95% CI, 44.4–97.5%) versus 0.0% (95% CI, 0.0–36.9%) in the test set (p = 0.0011). The wp-scores were also significantly associated with PFS in patients receiving single-agent ICI treatment (p < 0.05). In addition, the 5hmC predictive signature demonstrated superior predictive capability to tumor programmed death-ligand 1 and specificity to ICI treatment response prediction. Moreover, we identified novel 5hmC-associated genes and signaling pathways integral to ICI treatment response in lung cancer. This study provides proof-of-concept evidence that the cfDNA 5hmC signature is a robust biomarker for predicting ICI treatment response in lung cancer.
Full article
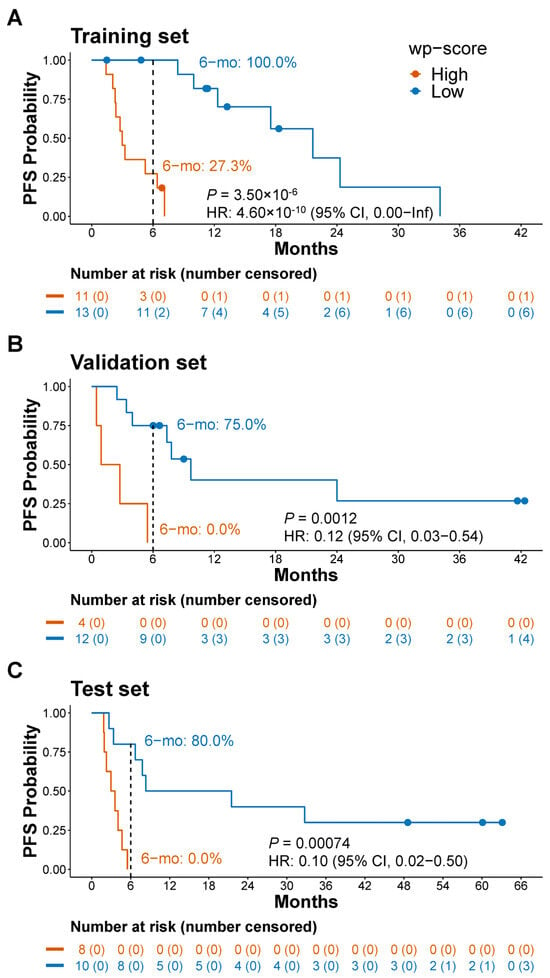
Figure 1
Open AccessCorrection
Correction: Won et al. Ex Vivo Perfusion Using a Mathematical Modeled, Controlled Gas Exchange Self-Contained Bioreactor Can Maintain a Mouse Kidney for Seven Days. Cells 2022, 11, 1822
by
Natalie Won, Jorge Castillo-Prado, Xinzhu Tan, John Ford, David Heath, Laura Ioana Mazilescu, Markus Selzner and Ian M. Rogers
Cells 2024, 13(8), 714; https://doi.org/10.3390/cells13080714 - 19 Apr 2024
Abstract
In the original publication [...]
Full article
Open AccessFeature PaperReview
Nuclear Phospholipids and Signaling: An Update of the Story
by
Irene Casalin, Eleonora Ceneri, Stefano Ratti, Lucia Manzoli, Lucio Cocco and Matilde Y. Follo
Cells 2024, 13(8), 713; https://doi.org/10.3390/cells13080713 - 19 Apr 2024
Abstract
In the last three decades, the presence of phospholipids in the nucleus has been shown and thoroughly investigated. A considerable amount of interest has been raised about nuclear inositol lipids, mainly because of their role in signaling acting. Here, we review the main
[...] Read more.
In the last three decades, the presence of phospholipids in the nucleus has been shown and thoroughly investigated. A considerable amount of interest has been raised about nuclear inositol lipids, mainly because of their role in signaling acting. Here, we review the main issues of nuclear phospholipid localization and the role of nuclear inositol lipids and their related enzymes in cellular signaling, both in physiological and pathological conditions.
Full article
(This article belongs to the Special Issue Nuclear Organization, Dynamics and Phase Separation in Health and Disease)
►▼
Show Figures
Figure 1
Open AccessReview
Hematopoietic Stem Cells as an Integrative Hub Linking Lifestyle to Cardiovascular Health
by
Xinliang Chen, Chaonan Liu, Junping Wang and Changhong Du
Cells 2024, 13(8), 712; https://doi.org/10.3390/cells13080712 - 19 Apr 2024
Abstract
Despite breakthroughs in modern medical care, the incidence of cardiovascular disease (CVD) is even more prevalent globally. Increasing epidemiologic evidence indicates that emerging cardiovascular risk factors arising from the modern lifestyle, including psychosocial stress, sleep problems, unhealthy diet patterns, physical inactivity/sedentary behavior, alcohol
[...] Read more.
Despite breakthroughs in modern medical care, the incidence of cardiovascular disease (CVD) is even more prevalent globally. Increasing epidemiologic evidence indicates that emerging cardiovascular risk factors arising from the modern lifestyle, including psychosocial stress, sleep problems, unhealthy diet patterns, physical inactivity/sedentary behavior, alcohol consumption, and tobacco smoking, contribute significantly to this worldwide epidemic, while its underpinning mechanisms are enigmatic. Hematological and immune systems were recently demonstrated to play integrative roles in linking lifestyle to cardiovascular health. In particular, alterations in hematopoietic stem cell (HSC) homeostasis, which is usually characterized by proliferation, expansion, mobilization, megakaryocyte/myeloid-biased differentiation, and/or the pro-inflammatory priming of HSCs, have been shown to be involved in the persistent overproduction of pro-inflammatory myeloid leukocytes and platelets, the cellular protagonists of cardiovascular inflammation and thrombosis, respectively. Furthermore, certain lifestyle factors, such as a healthy diet pattern and physical exercise, have been documented to exert cardiovascular protective effects through promoting quiescence, bone marrow retention, balanced differentiation, and/or the anti-inflammatory priming of HSCs. Here, we review the current understanding of and progression in research on the mechanistic interrelationships among lifestyle, HSC homeostasis, and cardiovascular health. Given that adhering to a healthy lifestyle has become a mainstream primary preventative approach to lowering the cardiovascular burden, unmasking the causal links between lifestyle and cardiovascular health from the perspective of hematopoiesis would open new opportunities to prevent and treat CVD in the present age.
Full article
(This article belongs to the Special Issue Stem Cell, Differentiation, Regeneration and Diseases)
►▼
Show Figures
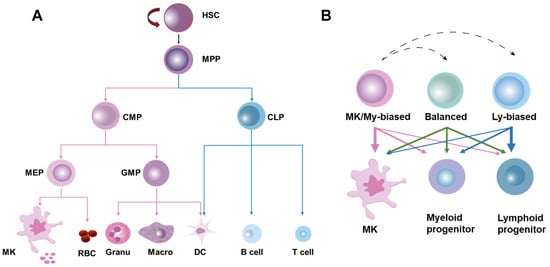
Figure 1
Open AccessArticle
Involvement of Mast Cells in the Pathology of COVID-19: Clinical and Laboratory Parallels
by
Andrey V. Budnevsky, Sergey N. Avdeev, Djuro Kosanovic, Evgeniy S. Ovsyannikov, Inessa A. Savushkina, Nadezhda G. Alekseeva, Sofia N. Feigelman, Viktoria V. Shishkina, Andrey A. Filin, Dmitry I. Esaulenko and Inna M. Perveeva
Cells 2024, 13(8), 711; https://doi.org/10.3390/cells13080711 - 19 Apr 2024
Abstract
►▼
Show Figures
Recent studies suggested the potential role of mast cells (MCs) in the pathology of coronavirus disease 2019 (COVID-19). However, the precise description of the MCs’ activation and the engagement of their proteases is still missing. The objective of this study was to further
[...] Read more.
Recent studies suggested the potential role of mast cells (MCs) in the pathology of coronavirus disease 2019 (COVID-19). However, the precise description of the MCs’ activation and the engagement of their proteases is still missing. The objective of this study was to further reveal the importance of MCs and their proteases (chymase, tryptase, and carboxypeptidase A3 (CPA3)) in the development of lung damage in patients with COVID-19. This study included 55 patients who died from COVID-19 and 30 controls who died from external causes. A histological analysis of the lung parenchyma was carried out to assess the protease profiles and degranulation activity of MCs. In addition, we have analyzed the general blood test, coagulogram, and C-reactive protein. The content of tryptase-positive MCs (Try-MCs) in the lungs of patients with COVID-19 was higher than in controls, but their degranulation activity was lower. The indicators of chymase-positive MCs (Chy-MCs) were significantly lower than in the controls, while the content of CPA3-positive MCs (CPA3-MCs) and their degranulation activity were higher in patients with COVID-19. In addition, we have demonstrated the existence of correlations (positive/negative) between the content of Try-MCs, Chy-MCs, and CPA3-MCs at different states of their degranulation and presence (co-adjacent/single) and the levels of various immune cells (neutrophils, eosinophils, basophils, and monocytes) and other important markers (blood hemoglobin, activated partial thromboplastin time (aPTT), international normalized ratio (INR), and fibrinogen). Thus, the identified patterns suggest the numerous and diverse mechanisms of the participation of MCs and their proteases in the pathogenesis of COVID-19, and their impact on the inflammatory process and coagulation status. At the same time, the issue requires further study in larger cohorts of patients, which will open up the possibility of using drugs acting on this link of pathogenesis to treat lung damage in patients with COVID-19.
Full article
Figure 1
Open AccessArticle
Genomic Engineering of Oral Keratinocytes to Establish In Vitro Oral Potentially Malignant Disease Models as a Platform for Treatment Investigation
by
Leon J. Wils, Marijke Buijze, Marijke Stigter-van Walsum, Arjen Brink, Britt E. van Kempen, Laura Peferoen, Elisabeth R. Brouns, Jan G. A. M. de Visscher, Erik H. van der Meij, Elisabeth Bloemena, Jos B. Poell and Ruud H. Brakenhoff
Cells 2024, 13(8), 710; https://doi.org/10.3390/cells13080710 - 19 Apr 2024
Abstract
Precancerous cells in the oral cavity may appear as oral potentially malignant disorders, but they may also present as dysplasia without visual manifestation in tumor-adjacent tissue. As it is currently not possible to prevent the malignant transformation of these oral precancers, new treatments
[...] Read more.
Precancerous cells in the oral cavity may appear as oral potentially malignant disorders, but they may also present as dysplasia without visual manifestation in tumor-adjacent tissue. As it is currently not possible to prevent the malignant transformation of these oral precancers, new treatments are urgently awaited. Here, we generated precancer culture models using a previously established method for the generation of oral keratinocyte cultures and incorporated CRISPR/Cas9 editing. The generated cell lines were used to investigate the efficacy of a set of small molecule inhibitors. Tumor-adjacent mucosa and oral leukoplakia biopsies were cultured and genetically characterized. Mutations were introduced in CDKN2A and TP53 using CRISPR/Cas9 and combined with the ectopic activation of telomerase to generate cell lines with prolonged proliferation. The method was tested in normal oral keratinocytes and tumor-adjacent biopsies and subsequently applied to a large set of oral leukoplakia biopsies. Finally, a subset of the immortalized cell lines was used to assess the efficacy of a set of small molecule inhibitors. Culturing and genomic engineering was highly efficient for normal and tumor-adjacent oral keratinocytes, but success rates in oral leukoplakia were remarkably low. Knock-out of CDKN2A in combination with either the activation of telomerase or knock-out of TP53 seemed a prerequisite for immortalization. Prolonged culturing was accompanied by additional genetic aberrations in these cultures. The generated cell lines were more sensitive than normal keratinocytes to small molecule inhibitors of previously identified targets. In conclusion, while very effective for normal keratinocytes and tumor-adjacent biopsies, the success rate of oral leukoplakia cell culturing methods was very low. Genomic engineering enabled the prolonged culturing of OL-derived keratinocytes but was associated with acquired genetic changes. Further studies are required to assess to what extent the immortalized cultures faithfully represent characteristics of the cells in vivo.
Full article
(This article belongs to the Special Issue Oral Diseases: Biological and Molecular Pathogenesis)
►▼
Show Figures
Figure 1

Journal Menu
► ▼ Journal Menu-
- Cells Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, Cells, IJMS, Life, Oxygen
Oxidative Stress and Inflammation, 2nd Volume
Topic Editors: Mohamad Allaw, Ines Castangia, Maria Letizia Manca, Matteo Perra, Amparo NacherDeadline: 31 May 2024
Topic in
Cancers, Cells, JCM, Radiation, Pharmaceutics, Applied Sciences, Nanomaterials, Current Oncology
Innovative Radiation Therapies
Topic Editors: Gérard Baldacchino, Eric Deutsch, Marie Dutreix, Sandrine Lacombe, Erika Porcel, Charlotte Robert, Emmanuelle Bourneuf, João Santos Sousa, Aurélien de la LandeDeadline: 30 June 2024
Topic in
Cells, Diseases, Healthcare, IJMS, Vaccines
Inflammation: The Cause of all Diseases 2.0
Topic Editors: Vasso Apostolopoulos, Jack Feehan, Vivek P. ChavdaDeadline: 31 July 2024
Topic in
Applied Sciences, Cancers, Cells, Electronics, AI
Explainable AI for Health
Topic Editors: Yudong Zhang, Juan Manuel Gorriz, Zhengchao DongDeadline: 8 August 2024

Conferences
Special Issues
Special Issue in
Cells
Islet Transplantation
Guest Editor: Shinichi MatsumotoDeadline: 26 April 2024
Special Issue in
Cells
Structures, Regulation, Signaling, and Physiological Functions of Adhesion G-Protein Coupled Receptors
Guest Editors: Ines Liebscher, Simone PrömelDeadline: 30 April 2024
Special Issue in
Cells
Microglia in Neurological Diseases
Guest Editors: Kate Lykke Lambertsen, Bente FinsenDeadline: 15 May 2024
Special Issue in
Cells
Cellular Reprogramming during Sepsis and Potential Therapeutic Targets
Guest Editor: Brendon SciclunaDeadline: 31 May 2024
Topical Collections
Topical Collection in
Cells
Role of Autophagy in Viral Infection
Collection Editor: Grant R. Campbell
Topical Collection in
Cells
Tumor Metabolism and Therapy
Collection Editors: Guohui Sun, Jianhua Wang
Topical Collection in
Cells
Molecular signaling, Circuit Neuroplasticity and the Cognitive Function
Collection Editor: Francisco Monje